Modernizing Trial Infrastructure: EDC Solution for CliniExperts
Modernizing Trial Infrastructure: EDC Solution for CliniExperts
In the world of clinical research, data integrity and regulatory readiness are mission-critical. For a growing Contract Research Organization (CRO) like CliniExperts, supporting global pharmaceutical and biotech clients, scaling operations meant rethinking how clinical data was managed, secured, and accessed across multiple studies.
To build a stronger, more scalable digital foundation, CliniExperts partnered with Tacten to design and implement a custom Electronic Data Capture (EDC) platform—tailored to meet the rigorous demands of modern trials and global regulatory standards.
Built for Trust: Why CliniExperts Chose Tacten Tacten was selected for our deep expertise in building healthcare-grade digital platforms that balance regulatory compliance with operational agility. With specialized experience in healthcare software, cloud automation, and Frappe-based systems, we brought the right mix of speed, control, and engineering precision. Our ability to bridge complex clinical workflows with secure, scalable infrastructure made us a trusted partner in this transformation.
The Challenge: Supporting Scalable, Compliant Growth
As the number and complexity of studies grew, so did the need for real-time visibility, consistent workflows, and reliable data traceability. CliniExperts needed a solution that could standardize how data was managed across multiple trials—without introducing risk or overhead.
Their ideal platform had to:
- Provide continuous monitoring, not just periodic reviews.
- Support parallel studies without cross-contamination.
- Maintain audit readiness at every stage of the study lifecycle.
- Offer secure access for globally distributed research teams.
Understanding the Context: Randomized Controlled Trials (RCTs)
Many clinical studies conducted by CROs like CliniExperts follow a Randomized Controlled Trial (RCT) design — widely regarded as the gold standard for evaluating new medical treatments. In an RCT, participants are randomly assigned to either a treatment group receiving the intervention or a control group receiving a placebo or standard care. Because the assignment is random, both groups are comparable except for the intervention itself. Researchers then compare outcomes between the groups, allowing any meaningful differences to be attributed to the treatment rather than external factors. This structured comparison makes RCTs highly reliable for establishing cause and effect and underscores the need for secure, traceable, and protocol-driven data systems.
The Objective: A Future-Ready Digital Foundation
Tacten’s mission was to deliver a robust, scalable EDC system that could serve as the digital backbone for CliniExperts’ trial operations. This included everything from configuring study templates and automating site setup to enforcing access control and ensuring full traceability—while keeping the system flexible enough to adapt to unique study needs.
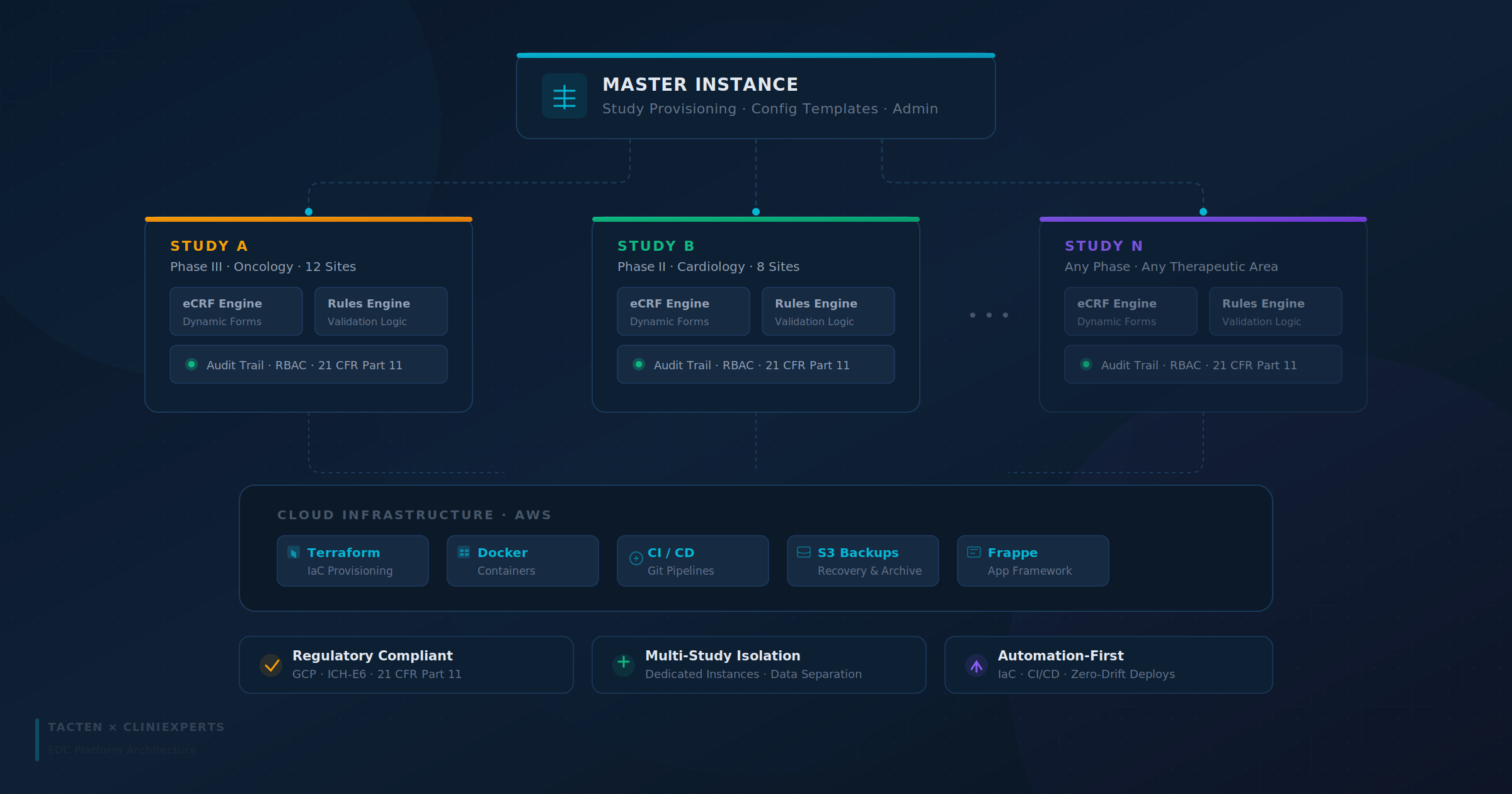
The Solution Stack: A Frappe-Powered Architecture
Tacten recognized that the Frappe Framework provided the perfect metadata-driven architecture for a configurable EDC. By combining Frappe’s robust backend with custom rule engines and isolated cloud deployments, we created a powerhouse solution.
Key Pillars of the Solution:
- Configurable Electronic Case Report Form (eCRF) Engine: A flexible data model allowing administrators to design and modify attributes dynamically, adapting to evolving study protocols in real-time.
- The Logic Center (Rules Engine): A sophisticated engine for data validation and workflow automation, ensuring that only "clean" data enters the system.
- Isolated Study Instances: Each clinical study operates within its own dedicated environment, ensuring strict data separation and governance.
- Compliance-First Design: Implemented deep-level audit trails and Role-Based Access Control (RBAC) to meet the rigorous standards of regulatory bodies.
Infrastructure Automation & DevOps Excellence
A key differentiator of this platform was its automation-first cloud architecture, engineered to eliminate manual infrastructure dependencies.
Built on AWS using Terraform-based Infrastructure as Code (IaC), every environment — Master, Study, Dev, UAT, and Production — can be provisioned through version-controlled templates, eliminating configuration drift and strengthening audit governance.
Core automation capabilities include:
- Master-Controlled Study Provisioning: Administrators can initiate a new study from a central instance, triggering automated deployment of a fully isolated Frappe site on a dedicated server with database setup and backup scheduling.
- CI/CD Pipelines: Structured Git-based workflows ensure controlled and traceable application releases.
- Containerized Deployments: Docker guarantees environment parity across development and production.
- Version-Governed Infrastructure: All infrastructure changes are managed as code, enabling rollback safety.
- Automated Backup & Recovery: Scheduled S3-backed backups protect study data across environments.
This engineering approach enabled CliniExperts to scale concurrent trials without increasing operational risk.
The Result: Faster Study Startups, Stronger Oversight
CliniExperts now manages their clinical trial infrastructure with greater agility and control. Studies can be launched faster, audits can be passed with confidence, and every stakeholder—from data managers to sponsors—has the real-time visibility they need to move forward.
Biograph is a clinical platform stack designed for teams building healthcare SaaS products. If you're evaluating foundations for your next health-tech product, let's talk.
No comments yet. Login to start a new discussion Start a new discussion